

French researchers made the discovery by analyzing blood from denim sandblasters. The results were presented at the Inflamma symposium organized by the Center for Research on Inflammatory Diseases in Brazil (image: Nature Communications)
Published on 05/12/2021
By Maria Fernanda Ziegler in Ribeirão Preto (Brazil) | Agência FAPESP – Researchers at the University of Orléans in France have discovered a key mechanism that activates lung inflammation caused by silicosis. This finding paves the way for new types of treatment for this disease.
Silicosis is an incurable lung disease caused by inhaling microcrystals of silica, a mineral present in sand, rock and mineral ores such as quartz. This disease mostly affects workers in mines, glass manufacturing and foundries. Silica is indestructible and eventually causes the death of several lung cell types, including macrophages, neutrophils, fibroblasts, and epithelial cells.
The resulting inflammation may be severe enough that lung tissue is entirely replaced with scar tissue by fibrosis, eventually leading to respiratory failure. Such patients will die without a lung transplant.
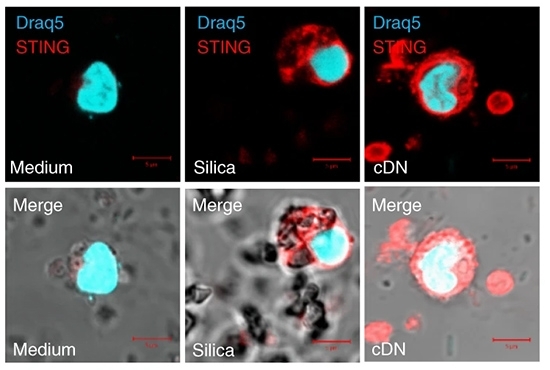
Experiments with mice exposed to silica showed that the inflammatory process and ensuing cell death trigger DNA leakage from cells.
“DNA released into the airways [because of cell death] activates cytosolic DNA sensors and the STING [stimulator of interferon genes] signaling pathway, which induces lung inflammation. This marks the onset of silicosis,” said Valérie Quesniaux, a researcher at the University of Orléans, in a presentation delivered to the Fifth International Symposium on Inflammatory Diseases (Inflamma) held in June 2019 at Ribeirão Preto, São Paulo State, by the Center for Research on Inflammatory Diseases (CRID), one of the Research, Innovation and Dissemination Centers (RIDCs) funded by FAPESP.
In an article published in Nature Communications, the researchers report that STING activation can be blocked by treatment with DNase I, an enzyme that degrades DNA released into the airways, thereby preventing intensification of the inflammation.
DNase I is used in the treatment of cystic fibrosis, a rare disease that affects the cells that produce mucus, sweat and digestive juices, thus causing these fluids to become thick and sticky such that they block tubes, ducts and passageways. A part of the study conducted in collaboration with researchers at Atatürk University’s Medical School in Turkey focused on analyzing blood plasma and lung secretions from sandblaster operators in denim factories.
“These workers were only 20 or so and already had silicosis as severe as the condition can become in much older men who have worked in mines all their lives. It was very striking indeed. They handled sandblasters without any kind of protection to make jeans look ‘distressed’, as the saying goes in the trade,” Quesniaux told Agência FAPESP.
In these patients, researchers observed not only a rise in DNA leakage but also an increased presence of inflammatory markers in blood and sputum. Analysis of their lung tissue showed high levels of STING pathway activation.
Relationship with tuberculosis
The group published another article in May 2019 in Cell Reports showing that lung inflammation induced by silica impairs tuberculosis control by affected individuals. According to the study, silica exacerbates infection by Mycobacterium tuberculosis, the bacillus that causes tuberculosis.
“Silica-induced inflammation contributes to the development of tuberculosis. We showed that exposure to silica triggers a type 2 immune response, which isn’t an effective defense against tuberculosis. This happens because prior exposure to silica boosts pulmonary Th2 cell and M2 macrophage responses, reducing the chance of an effective type 1 immune response,” Quesniaux said.
Type 1 immune responses involving pro-inflammatory molecules such as interferon-gamma and TNF and TH1 cells are essential to control M. tuberculosis infection.
“Prior silica exposure, however, means several of these molecules have already been recruited by the immune system and are working against silicosis. This forces a type 2 immune response to tuberculosis, which is far better for the bacillus than for the host,” Quesniaux said.
“We obtained interesting results on the silicosis mechanism and its relationship to tuberculosis. However, it hasn’t yet been explained to the world, and especially to young people, that distressed jeans are produced by this hazardous sandblasting process in tiny workshops where the workers wear no protective equipment.”
The article “STING-dependent sensing of self-DNA drives silica-induced lung inflammation” (doi: 10.1038/s41467-018-07425-1) by Sulayman Benmerzoug, Stéphanie Rose, Badreddine Bounab, David Gosset, Laure Duneau, Pauline Chenuet, Lucile Mollet, Marc Le Bert, Christopher Lambers, Silvana Geleff, Michael Roth, Louis Fauconnier, Delphine Sedda, Clarisse Carvalho, Olivier Perche, David Laurenceau, Bernhard Ryffel, Lionel Apetoh, Ahmet Kiziltunc, Hakan Uslu, Fadime Sultan Albez, Metin Akgun, Dieudonnée Togbe and Valerie F. J. Quesniaux can be read at www.nature.com/articles/s41467-018-07425-1.
The article “Sterile lung inflammation induced by silica exacerbates Mycobacterium tuberculosis infection via STING-dependent type 2 immunity” (doi: 10.1016/j.celrep.2019.04.110) by Sulayman Benmerzoug, Badreddine Bounab, Stéphanie Rose, David Gosset, Franck Biet, Thierry Cochard, Aurore Xavier, Nathalie Rouxel, Louis Fauconnier, William G.C. Horsnell, Bernhard Ryffel, Dieudonnée Togbe and Valerie F.J. Quesniaux can be read at www.cell.com/cell-reports/fulltext/S2211-1247(19)30600-X.
Source: https://agencia.fapesp.br/30974