

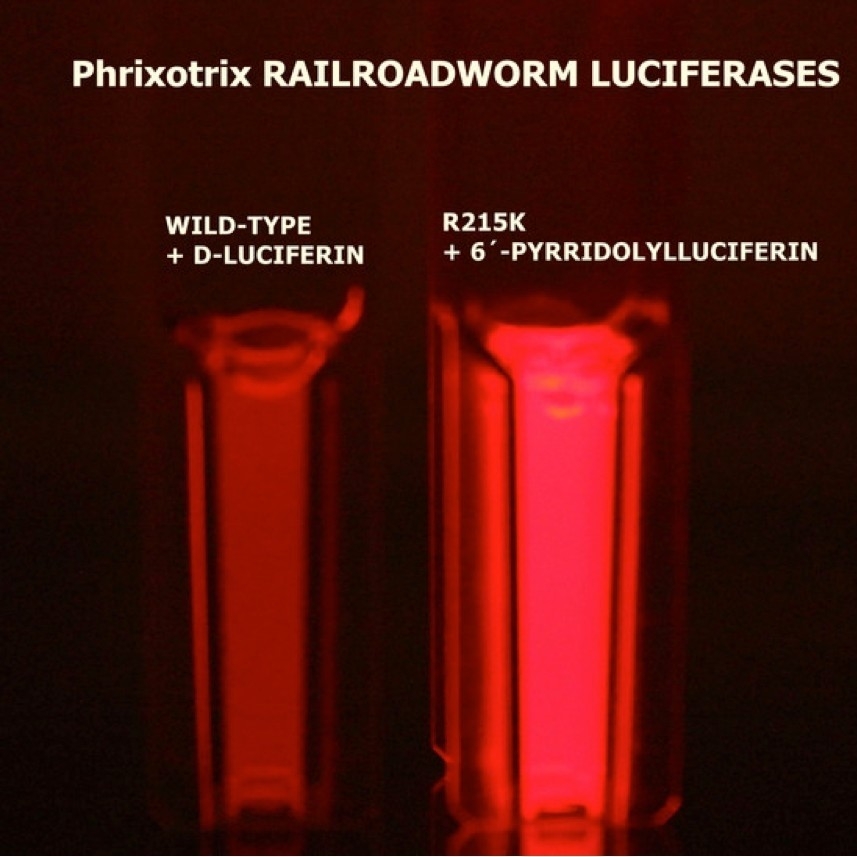
Luciferin-luciferase system developed in collaboration with Japanese researchers produces brighter and longer-lasting far red light. The innovation can be used to image cells and tissues for diagnosis and biomedical research (illustration of the brighter bioluminescence obtained by the UFSCar team – right-hand column; credit: International Journal of Molecular Sciences)
Published on 04/01/2021
By José Tadeu Arantes | Agência FAPESP – Researchers at the Federal University of São Carlos (UFSCar) in the state of São Paulo, Brazil, have developed a novel far red light-emitting luciferin-luciferase system that is more efficient than those available commercially. An article on the subject is published in the International Journal of Molecular Sciences.
The study was supported by FAPESP via the Thematic Project “Arthropod bioluminescence: biological diversity in Brazilian biomes, biochemical origin, structural/functional evolution of luciferases, molecular differentiation of lanterns, biotechnological, environmental and educational applications”, for which the principal investigator is Vadim Viviani, a biochemist and professor at UFSCar.
“We obtained a novel luciferin-luciferase system that produces far red light at the wavelength of 650 nanometers and emits the brightest bioluminescence ever reported in this part of the spectrum. It’s a highly promising result for bioluminescence imaging of biological and pathological processes in mammalian tissues,” Viviani told Agência FAPESP.
Luciferases are enzymes that catalyze the oxidation of luciferins, compounds present in some animals, algae and fungi. The oxidation reaction is responsible for the phenomenon of bioluminescence, which consists of the emission of light at wavelengths ranging from blue to red.
The firefly’s luciferin-luciferase system is widely used to help produce images of cell cultures and live animal models. It helps physicians monitor metastasis, for example, and see how tumors respond to treatment. It is also used to follow the viral infection process and the effects of candidate drugs on viruses, including the novel coronavirus.
“Red bioluminescence is preferred when imaging biological or pathological processes in mammalian tissues because hemoglobin, myoglobin and melanin absorb little long-wavelength light. Detection is best of all in the far red and near-infrared bands, but bioluminescent systems that naturally emit far red light don’t exist,” Viviani said.
“Some genetically modified forms of luciferase and synthetic analogs of natural luciferins are produced commercially. In conjunction, they produce light at wavelengths as long as 700 nanometers, but the light produced by these artificial systems is generally much weaker and more short-lived than light from natural bioluminescent systems.”
Viviani and collaborators used genetic engineering to modify luciferase from the Railroad worm Phrixothrix hirtus, the only luciferase that naturally emits red light, cloned by Viviani two decades ago. This they combined with luciferin analogs synthesized by colleagues at the University of Electro-Communications in Tokyo, Japan. The result was a much more efficient far red luciferin-luciferase system.
“Our best combination produces far red at 650 nanometers, three times brighter than natural luciferin and luciferase, and roughly 1,000 times brighter than the same luciferase with a commercial analog,” Viviani said.
“Besides the long wavelength and intense brightness, our combination has better thermal stability and cell membrane penetrability. Above all, it produces more lasting continuous bioluminescence, taking at least an hour to decay and significantly facilitating the real-time imaging of biological and pathological processes.”
The project was a continuation of a PhD thesis recently defended by Vanessa Bevilaqua on the effect of synthetic luciferin analogs on red luciferase from Phrixothrix. The other co-authors are PhD candidate Daniel Sousa, Gabriel Pelentir with a scientific initiation grant, and Michio Kakiuchi and Takashi Hirano in Japan.
The article “A very bright far red bioluminescence emitting combination based on engineered railroad worm luciferase and 6′-amino-analogs for bioimaging purposes” is at: www.mdpi.com/1422-0067/22/1/303.
Source: https://agencia.fapesp.br/35516