

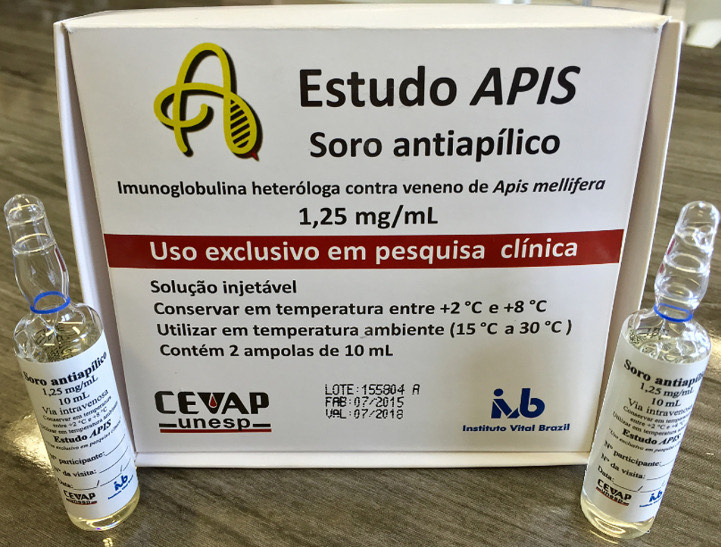
The next step is to conduct the third and final phase of clinical trials with 150 to 200 patients (photo: CTS-CEVAP)
Published on 03/05/2026
Agência FAPESP* – Researchers at São Paulo State University (UNESP) in Brazil have received BRL 20 million from the Ministry of Health to begin the final phase of testing for an anti-bee serum that treats poisoning by Africanized bees (Apis mellifera). The Center for Translational Science and Biopharmaceutical Development (CTS) will conduct the work. The CTS is a FAPESP Science Center for Development (SCD).
Using 100% Brazilian technology, scientists at the Center for the Study of Venoms and Venomous Animals (CEVAP) at UNESP’s Botucatu campus began developing the biopharmaceutical more than a decade ago. The project has received support from FAPESP.
Africanized bees, the result of crossbreeding between African and European bees, have become one of the most common species in Brazil. They are primarily responsible for honey production in Brazil, but they are also often involved in accidents with humans. Incidents range from mild stings, which cause only local pain and swelling, to severe systemic reactions. These occur because of sensitivity to venom components or multiple stings, which can lead to breathing difficulties, shock, tremors, kidney failure, and other symptoms.
The use of pesticides, deforestation, and the decline of floral resources have increased encounters with bees in urban environments. Between 2013 and 2023, Brazil recorded 206,746 cases of bee stings. In 2023 alone, there were 33,317 cases, surpassing the 32,420 accidents involving snakes. These incidents resulted in 649 direct and 50 indirect deaths. Thus, an ecosystem conducive to a neglected public health problem has been created: a potential increase in cases coupled with the lack of adequate treatment.
“Unfortunately, we don’t currently have a specific antidote for these accidents in the healthcare system. Treatment with drugs to control pain, inflammation, and allergies is the standard protocol performed in any hospital. However, the lack of a specific treatment makes these cases more serious and can lead to death,” points out Rui Seabra Ferreira Jr., executive coordinator of CEVAP.

In 2023, there were 33,317 bee-related accidents, surpassing the 32,420 snake-related accidents (photo: Letícia Alcântara/iNaturalist)
The bioproduct began production in 2009 after Ferreira Jr.’s graduate and postdoctoral studies evaluated its concept. Like other serums, the anti-apiculum serum is produced by gradually inoculating Africanized bee venom into horses to stimulate antibody production in the blood plasma. The blood is collected, the plasma is purified, and the serum is formulated.
In 2013, Benedito Barraviera led CEVAP in conducting a phase 1/2 multicenter clinical trial in partnership with researchers from the Butantan Institute and the Vital Brazil Institute in the state of Rio de Janeiro. The study was conducted at the general and teaching hospital (“Hospital das Clínicas”) of the UNESP School of Medicine in Botucatu and at the Nossa Senhora da Conceição Hospital in Tubarão, Santa Catarina state, from 2016 to 2018. It proved the safety and preliminary efficacy of the serum. Twenty adult volunteers, with an average age of 44, participated in the study. They had been exposed to a number of stings ranging from seven to 2,000. No serious adverse effects were reported, and all patients showed clinical improvement. In early 2023, the National Institute of Industrial Property (INPI) granted the patent for the anti-bee venom serum.
The next step is to conduct the third and final phase of clinical trials with 150 to 200 patients. Approval of funding under the Ministry of Health’s 2024 Local Development and Innovation Program (PDIL) call for proposals in September will enable the final phase of serum testing. Afterwards, it will be possible to apply for registration of the drug with the Brazilian Health Regulatory Agency (ANVISA) and obtain authorization for commercial production and inclusion in the Unified Health System (SUS), the national public health network.
* With information from Juliana Marques from the CTS-CEVAP
Source: https://agencia.fapesp.br/57375