

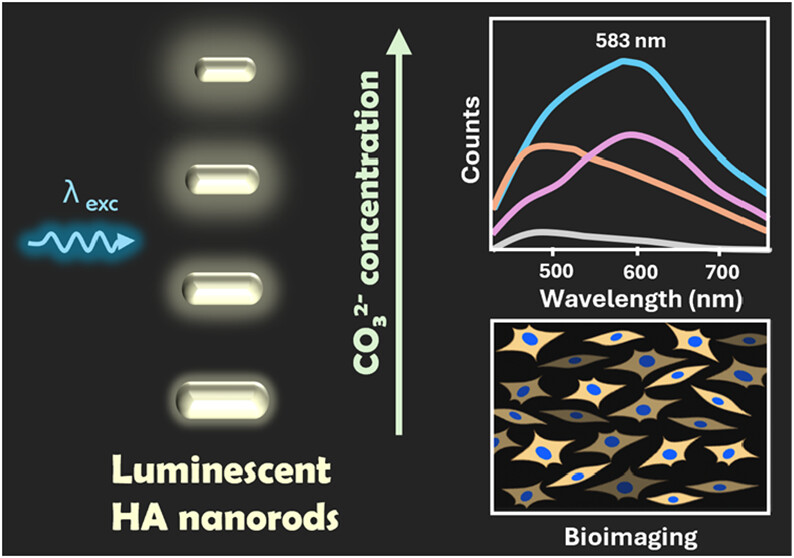
By adding carbonate groups during hydroxyapatite synthesis, the intrinsic luminescence of the material is intensified through an increase in the concentration of crystalline defects in its structure (image: ACS Nanoscience Au)
Published on 03/16/2026
Agência FAPESP – The Nanomedicine and Nanotoxicology Group (GNano) at the University of São Paulo’s São Carlos Institute of Physics (IFSC-USP) in Brazil has discovered a way to transform hydroxyapatite, a bioceramic material, into a nanoparticle with enhanced intrinsic luminescence. This paves the way for the use of biocompatible, low-cost nanomaterials in biomedical imaging techniques.
“We’ve demonstrated that the incorporation of carbonate groups into the hydroxyapatite structure increases the concentration of crystal defects, which are responsible for enhancing the intrinsic luminescence of the material. After functionalization with citrate, which improves colloidal stability in aqueous media, these calcium phosphate nanoparticles can be used as luminescent agents for cellular bioimaging,” Thales Rafael Machado, one of the participants in the study and a FAPESP scholarship recipient, explained to Agência FAPESP.
Professor Valtencir Zucolotto of the IFSC-USP coordinated the research, which was conducted in collaboration with the Center for Molecular Engineering of Advanced Materials (CEMol), a FAPESP Research, Innovation and Dissemination Center (RIDC) headquartered at the Brazilian Center for Research in Energy and Materials (CNPEM) in Campinas.
According to Machado, the findings were achieved by controlling the concentration of structural and point defects in hydroxyapatite nanoparticles by incorporating different levels of carbonate during synthesis. The sample with the highest carbonate content exhibited the most intense luminescence.
“The bioimaging capability was demonstrated by visualizing the internalization of the nanoparticles into cells using confocal fluorescence microscopy, relying exclusively on their intrinsic luminescence. Cellular internalization was also confirmed by flow cytometry, likewise based on the luminescent signal of the particles, while biocompatibility was assessed through cellular cytotoxicity assays,” says Machado.
According to Machado, studying defect chemistry and intrinsic luminescence in carbonated hydroxyapatite contributes to developing new hydroxyapatite-based photocatalytic materials for environmental applications. It also provides a foundation for spectroscopic studies of hard tissues, such as bones and teeth. “This knowledge can also be applied to producing luminescent scaffolds for tissue engineering,” he adds.
The study was published in the journal ACS Nanoscience Au from the American Chemical Society.
Cancer treatment
In a parallel study, GNano and CEMol developed an efficient, robust strategy for delivering gemcitabine – a widely used chemotherapy drug for cancers such as pancreatic cancer – via calcium phosphate nanoparticles.
“The system was designed to be dual pH-responsive, keeping the drug inactive under normal physiological conditions, such as in the bloodstream, and releasing the drug in its active form only in more acidic environments, characteristic of tumor regions. This promotes greater bioavailability and therapeutic potential,” says Machado.
The study was published in ACS Applied Bio Materials, also from the American Chemical Society.
Additionally, the group demonstrated the ability to functionalize the surface of the nanoparticles with folic acid via highly stable covalent bonds. Folate acts as a targeting molecule because many tumor cells have a higher demand for this vitamin.
“Thus, the system combines controlled release and active targeting, promoting higher drug concentrations in tested tumor cells – from breast and cervical cancer – potentially reducing unwanted side effects in healthy tissues,” explains the USP São Carlos researcher.
The findings began with the development of a prodrug in which gemcitabine was linked to a biocompatible polymer called carboxymethylcellulose. In this form, the drug is better protected from premature degradation in the body and is only released in acidic environments, such as those found in tumors or certain cellular vesicles, while remaining stable in the bloodstream. The same polymer was also used to stabilize calcium phosphate nanoparticles in suspension and prevent agglomeration.
Machado emphasizes that these findings contribute to the development of safer and more effective cancer treatments. “By keeping the drug inactive while it circulates in the body and releasing it preferentially in the tumor environment, the system has the potential to reduce side effects and enhance the action of the drug directly on tumor cells.”
Using nanoparticles made of calcium phosphate – a biocompatible material that is naturally present in the human body – makes the strategy safer. The combination of controlled release, response to tumor pH, and active targeting via folic acid represents an advance in nanomedicine and more precise therapies.
“Systems like these could make chemotherapy more efficient with lower doses and less damage to healthy tissues, improving patients’ quality of life during treatment,” says the researcher.
GNano has developed a series of new nanostructured materials for advanced cancer diagnostics and therapies, as well as safer delivery systems for agricultural pesticides and biological inputs.
The article “Defect-related photoluminescence in hydroxyapatite nanoparticles modulated by carbonate incorporation” can be read at pubs.acs.org/doi/full/10.1021/acsnanoscienceau.5c00140.
The article “Dual pH-responsive calcium phosphate nanoparticles conjugated with folate by CuAAC click chemistry for targeted gemcitabine delivery to cancer cells” can be read at pubs.acs.org/doi/full/10.1021/acsabm.5c01683.
Source: https://agencia.fapesp.br/57479